Thujone Cas 76231-76-0
Thujone (/ˈθuːdʒoʊn/ ⓘ[2]) is a ketone and a monoterpene that occurs predominantly in two diastereomeric (epimeric) forms: (−)-α-thujone and (+)-β-thujone.[3][4]
Though it is best known as a chemical compound in the spirit absinthe, it is only present in trace amounts and is unlikely to be responsible for the spirit’s purported stimulant and psychoactive effects.[5][6][7]
Thujone acts on the GABAA receptor as an antagonist. As a competitive antagonist of GABAA receptor, thujone alone is considered to be convulsant,[8] though by interfering with the inhibitory transmitter GABA, it may convey stimulating, mood-elevating effects at low doses.[citation needed] It is also found in perfumery as a component of several essential oils.[citation needed]
In addition to the naturally occurring (−)-α-thujone and (+)-β-thujone, two other forms are possible: (+)-α-thujone and (−)-β-thujone. In 2016, they were found in nature as well,[9] in Salvia officinalis.
-
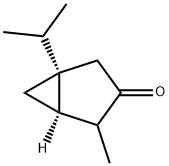
(−)-α-thujone
-
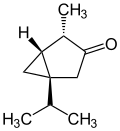
(+)-α-thujone
-
(+)-β-thujone
-
(−)-β-thujone
Sources
Thujone is found in a number of plants, such as arborvitae (genus Thuja, hence the derivation of the name), Nootka cypress, some junipers, mugwort, oregano, common sage (sage oil contain more than 20% from alpha and beta thujone), tansy, and wormwood, most notably grand wormwood (Artemisia absinthium), usually as a mix of isomers in a 1:2 ratio. It is also found in various species of Mentha (mint).
Biosynthesis
The biosynthesis of thujone is similar to the synthesis of other monoterpenes and begins with the formation of geranyl diphosphate (GPP) from dimethylallyl pyrophosphate (DMAPP) and isopentenyl diphosphate (IPP), catalyzed by the enzyme geranyl diphosphate synthase.[10] Quantitative 13C NMR spectroscopic analysis has demonstrated that the isoprene units used to form thujone in plants are derived from the methylerythritol phosphate pathway (MEP).[11]
The reactions that generate the thujone skeleton in sabinene from GPP are mediated by the enzyme sabinene synthase which has GPP as its substrate.[10] GPP (1) first isomerizes to linalyl diphosphate (LPP) (2) and neryl diphosphate (NPP) (3). LPP preferentially forms a delocalized allylic cation-diphosphate (4). The ion-pair intermediate then cyclizes in an electrophilic addition to yield the α-terpinyl tertiary cation (5).[10]

The α-terpinyl cation (5) then undergoes a 1,2 hydride shift via a Wagner–Meerwein rearrangement, leading to the formation of the terpinen-4-yl cation (6). This cation undergoes a second cyclization to form the thujyl cation intermediate (7) before loss of a proton to form the thujone precursor, (+)-sabinene (8).

From (+)-sabinene (8), the proposed biosynthetic route to generate thujone follows a three-step pathway: (+)-sabinene is first oxidized to an isomer of (+)-sabinol (9-1, 9-2) by a cytochrome P450 enzyme, followed by conversion to (+)-sabinone (10) via a dehydrogenase. Finally, a reductase mediates the conversion to α-thujone (11-1) and β-thujone (11-2).[12] The isomerism of the (+)-sabinol intermediate varies among thujone-producing plants; for instance, in the western redcedar (Thuja plicata), thujone is derived exclusively from the (+)-trans-sabinol intermediate (9-1) whereas in the common garden sage (Salvia officinalis), thujone is formed from the (+)-cis-sabinol intermediate (9-2).[13]




Reviews
There are no reviews yet.