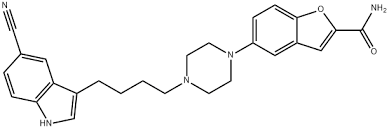
Buy Vilazodone (Viibryd) Cas 163521-12-8
Vilazodone, sold under the brand name Viibryd among others, is a medication used to treat major depressive disorder.[1] It is classified as a serotonin modulator[1] and is taken by mouth.[1]
Its common side effects include nausea, diarrhea, and trouble sleeping.[1] Serious side effects may include increased suicidal thoughts or actions in those under the age of 25, serotonin syndrome, bleeding, activation of mania or hypomania, pancreatitis, seizures, angle-closure glaucoma, sleep paralysis, and sexual dysfunction.[6]
Vilazodone may cause a syndrome of inappropriate antidiuretic hormone secretion (SIADH).[1] A withdrawal syndrome may occur if the dose is rapidly decreased.[1] Use during pregnancy and breastfeeding is not generally recommended.[7] It is in the serotonin modulator class of medications and is believed to work both as a selective serotonin reuptake inhibitor (SSRI) and activator of the 5-HT1A receptor.[1]
Vilazodone was approved for medical use in the United States in 2011[1] and in Canada in 2018.[8] In 2019, it was the 334th most commonly prescribed medication in the United States, with more than 900 thousand prescriptions.[9] The drug lost patent protection in June 2022 for adults and in July 2023 for pediatrics.[10] Generic versions have been approved by the US Food and Drug Administration.[11][12]
Medical uses
Seven controlled efficacy trials were conducted of vilazodone for treatment of major depressive disorder.[13] Five of these trials showed no significant influence of vilazodone over placebo on depressive symptoms.[13] In the remaining two trials, small but significant advantages of vilazodone over placebo were found.[13][14] According to these two eight-week trials in adults, vilazodone has an antidepressant response after one week of treatment.[15] After eight weeks it resulted in a 13% greater response than placebo.[15] Remission rates, however, were not significantly different versus placebo.[15]
According to the US Food and Drug Administration (FDA) in 2011, “it is unknown whether vilazodone has any advantages compared to other drugs in the antidepressant class.”[16] A 2019 review stated that “present studies do not suggest the superiority of vilazodone compared with other antidepressants.”[17]
Development of vilazodone for generalized anxiety disorder has been stopped as of 2017.[18] While there is tentative evidence of a small benefit in generalized anxiety disorder, there is a high rate of side effects.[19]
Adverse effects
In September 2016, the US Food and Drug Administration (FDA) required a new warning to be added to the prescribing information related to a link between vilazodone and acute pancreatitis and sleep paralysis.[20] In addition, other sleep disturbances such as hypnagogic hallucinations and sleep terrors can occur.[21][22]
Sleep paralysis is a state, during waking up or falling asleep, in which a person is conscious but experiences full-body paralysis. During an episode, the person may hallucinate (hear, feel, or see things that are not there), which often results in fear.[23] A night terror, also called sleep terror, is a sleep disorder causing feelings of panic or dread.[24] The rate of sleep paralysis adverse events was high enough to merit an FDA warning added to the Viibryd prescription label.[6][21]

Reviews
There are no reviews yet.