Buy Tryptamine (2-(3-indolyl)ethylamine) Cas 61-54-1
Buy Tryptamine (2-(3-indolyl)ethylamine) Cas 61-54-1
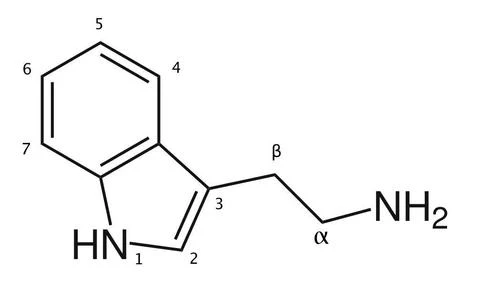
Tryptamine, also known as 2-(3-indolyl)ethylamine, is an indolamine metabolite of the essential amino acid tryptophan.[9][10] The chemical structure is defined by an indole—a fused benzene and pyrrole ring, and a 2-aminoethyl group at the second carbon (third aromatic atom, with the first one being the heterocyclic nitrogen).[9] The structure of tryptamine is a shared feature of certain aminergic neuromodulators including melatonin, serotonin, bufotenin and psychedelic derivatives such as dimethyltryptamine (DMT), psilocybin, psilocin and others.[11][12][13]
Tryptamine has been shown to activate serotonin receptors[5][14] and trace amine-associated receptors expressed in the mammalian brain, and regulates the activity of dopaminergic, serotonergic and glutamatergic systems.[15][16] In the human gut, bacteria convert dietary tryptophan to tryptamine, which activates 5-HT4 receptors and regulates gastrointestinal motility.[10][17][18]
Multiple tryptamine-derived drugs have been developed to treat migraines, while trace amine-associated receptors are being explored as a potential treatment target for neuropsychiatric disorders.[19][20][21]
Natural occurrences
For a list of plants, fungi and animals containing tryptamines, see List of psychoactive plants and List of naturally occurring tryptamines.
Mammalian brain
Endogenous levels of tryptamine in the mammalian brain are less than 100 ng per gram of tissue.[12][16] However, elevated levels of trace amines have been observed in patients with certain neuropsychiatric disorders taking medications, such as bipolar depression and schizophrenia.[22]
Mammalian gut microbiome
Tryptamine is relatively abundant in the gut and feces of humans and rodents.[10][17] Commensal bacteria, including Ruminococcus gnavus and Clostridium sporogenes in the gastrointestinal tract, possess the enzyme tryptophan decarboxylase, which aids in the conversion of dietary tryptophan to tryptamine.[10] Tryptamine is a ligand for gut epithelial serotonin type 4 (5-HT4) receptors and regulates gastrointestinal electrolyte balance through colonic secretions.[17]
Metabolism
Biosynthesis
To yield tryptamine in vivo, tryptophan decarboxylase removes the carboxylic acid group on the α-carbon of tryptophan.[12] Synthetic modifications to tryptamine can produce serotonin and melatonin; however, these pathways do not occur naturally as the main pathway for endogenous neurotransmitter synthesis.[23]
Catabolism
Monoamine oxidases A and B are the primary enzymes involved in tryptamine metabolism to produce indole-3-acetaldehyde, however it is unclear which isoform is specific to tryptamine degradation.[24]
Figure

Biological activity
| Target | Affinity (Ki, nM) | Species |
|---|---|---|
| 5-HT1A | 32–105 (Ki) 899–>10,000 (EC50Tooltip half-maximal effective concentration) ND (EmaxTooltip maximal efficacy) |
Human Human Human |
| 5-HT1B | 36–525 | Human |
| 5-HT1D | 23–521 | Human |
| 5-HT1E | 2,559 | Human |
| 5-HT1F | 2,409 | Human |
| 5-HT2A | 37–4,070 (Ki) 7.4–257 (EC50) 71–104% (Emax) |
Human Human Human |
| 5-HT2B | 25–113 (Ki) 29.5 (EC50) 92% (Emax) |
Human Human Human |
| 5-HT2C | 17–3,000 (Ki) 1.17–45.7 (EC50) 85–108% (Emax) |
Human Human Human |
| 5-HT3 | ND | ND |
| 5-HT4 | >10,000 13,500 (EC50) 96% (Emax) |
Mouse Pig Pig |
| 5-HT5A | ND | ND |
| 5-HT6 | 70–438 | Human |
| 5-HT7 | 148–158 | Human |
| α2A | 19,000 | Rat |
| TAAR1Tooltip Human trace amine-associated receptor 1 | 1,400 (Ki) 2,700 (EC50) 117% (Emax) 130 (Ki) 410 (EC50) 91% (Emax) 1,084 (Ki) 2,210–21,000 (EC50) 73% (Emax) |
Mouse Mouse Mouse Rat Rat Rat Human Human Human |
| SERTTooltip Serotonin transporter | 32.6 (EC50) a | Rat |
| NETTooltip Norepinephrine transporter | 716 (EC50) a | Rat |
| DATTooltip Dopamine transporter | 164 (EC50) a | Rat |
| Note: The smaller the value, the more avidly the compound binds to or activates the site. Footnotes: a = Neurotransmitter release. Refs: Main:[25][26] Additional:[27][28][29][5][30][31][32][33][34][35][36] | ||
Serotonin receptor agonist
Tryptamine is known to act as a serotonin receptor agonist, although its potency is limited by rapid inactivation by monoamine oxidases.[4][5][14][37][38][39] It has specifically been found to act as a full agonist of the serotonin 5-HT2A receptor (EC50Tooltip half-maximal effective concentration = 7.36 ± 0.56 nM; Emax = 104 ± 4%).[5] Tryptamine was of much lower potency in stimulating the 5-HT2A receptor β-arrestin pathway (EC50 = 3,485 ± 234 nM; Emax = 108 ± 16%).[5] In contrast to the 5-HT2A receptor, tryptamine was found to be inactive at the serotonin 5-HT1A receptor.[5]
Gastrointestinal motility
Tryptamine produced by mutualistic bacteria in the human gut activates serotonin GPCRs ubiquitously expressed along the colonic epithelium.[17] Upon tryptamine binding, the activated 5-HT4 receptor undergoes a conformational change which allows its Gs alpha subunit to exchange GDP for GTP, and its liberation from the 5-HT4 receptor and βγ subunit.[17] GTP-bound Gs activates adenylyl cyclase, which catalyzes the conversion of ATP into cyclic adenosine monophosphate (cAMP).[17] cAMP opens chloride and potassium ion channels to drive colonic electrolyte secretion and promote intestinal motility.[18][40]

Reviews
There are no reviews yet.