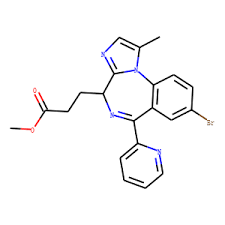
Buy Remimazolam (Byfavo) Cas 308242-62-8
Remimazolam, sold under the brand name Byfavo, is a medication for the induction and maintenance of procedural sedation in adults for invasive diagnostic or surgical procedures lasting 30 minutes or less.[2][5][6] It is a benzodiazepine drug, developed by PAION AG in collaboration with several regional licensees as an alternative to the short-acting imidazobenzodiazepine midazolam, for use in the induction of anesthesia and conscious sedation for minor invasive procedures.
Remimazolam was found to have both a more rapid onset and a shorter duration than midazolam, and human clinical trials showed a faster recovery time and predictable, consistent pharmacokinetics, suggesting some advantages over existing drugs for these applications.[7][8]
The most common side effects for procedural sedation include low blood pressure, high blood pressure, diastolic hypertension, systolic hypertension, low blood oxygen level, and diastolic hypotension.[5][6]
Remimazolam was approved for medical use in the United States in July 2020,[5][6] and in the European Union in March 2021.[3]
Medical uses
Remimazolam is indicated for the induction and maintenance of procedural sedation in adults (lasting 30 minutes or less in the US).[5][6][9]
History
Remimazolam was approved for medical use in the United States in July 2020.[5][6]
The U.S. Food and Drug Administration (FDA) approved remimazolam based on evidence from three clinical trials (Trial 1/NCT02290873, Trial 2/NCT02296892 and Trial 3/NCT02532647) in adults undergoing short procedures.[5] Trials were conducted at 32 sites in the United States.[5]
Trials 1 and 3 were conducted in participants undergoing colonoscopy and Trial 2 was conducted in participants undergoing bronchoscopy procedures.[5]
In the trials, participants were randomly divided in three groups: one group received remimazolam, one group received placebo and one group received midazolam (similar, but approved drug).[5] In the first two groups, neither participants nor investigators knew which medications were given and participants could also receive midazolam as a rescue drug when needed for sedation.[5] In the third group, all participants received midazolam only[5] Additionally, in all three trials participants received a medication for pain control[5]
Trials 1 and 2 compared participants who received remimazolam to participants in the other two groups, measuring the success of sedation with the set of pre-determined criteria.[5] Data from Trial 3 were used primarily to assess the side effects of remimazolam when multiple dosing is used.[5]

Reviews
There are no reviews yet.