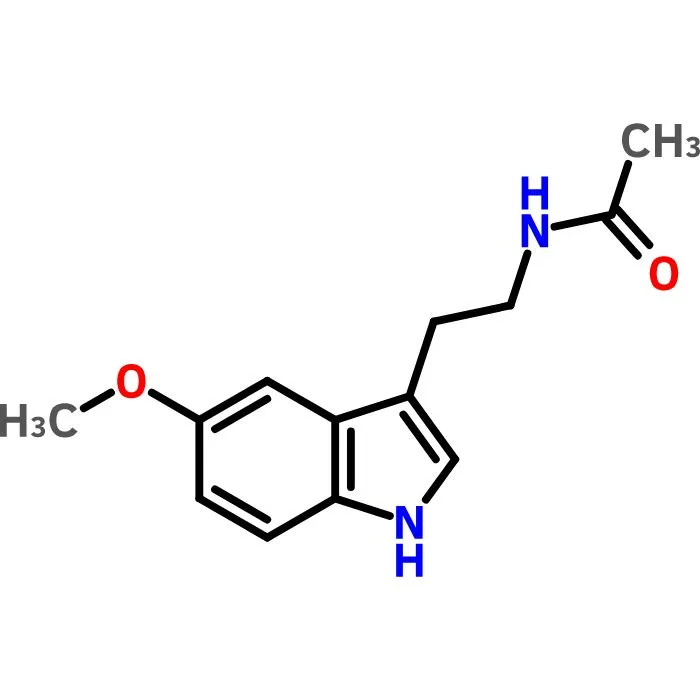
Buy Melatonin Cas 73-31-4
Buy Melatonin Cas 73-31-4
Melatonin, an indoleamine, is a natural compound produced by various organisms, including bacteria and eukaryotes.[1] Its discovery in 1958 by Aaron B. Lerner and colleagues stemmed from the isolation of a substance from the pineal gland of cows that could induce skin lightening in common frogs. This compound was later identified as a hormone secreted in the brain during the night, playing a crucial role in regulating the sleep-wake cycle, also known as the circadian rhythm, in vertebrates.[2][3]
In vertebrates, melatonin’s functions extend to synchronizing sleep-wake cycles, encompassing sleep-wake timing and blood pressure regulation, as well as controlling seasonal rhythmicity (circannual cycle), which includes reproduction, fattening, molting, and hibernation.[4] Its effects are mediated through the activation of melatonin receptors and its role as an antioxidant.[5][6][7] In plants and bacteria, it serves as a defense mechanism against oxidative stress, indicating its evolutionary significance.[8] Mitochondria, key organelles, are the main producers of melatonin,[9] underscoring its “ancient origins” and its fundamental role in protecting the earliest cells from reactive oxygen species.[10][11]
In addition to its endogenous functions as a hormone and antioxidant, melatonin is also administered exogenously as a dietary supplement and medication. Melatonin is used medically primarily for sleep-related problems: for example, prolonged-release melatonin (Circadin) is approved in several countries for short-term treatment of insomnia in people aged 55 years of age or older.[12] It is used in the treatment of sleep disorders, including insomnia and various circadian rhythm sleep disorders.
Biological activity
In humans, melatonin is presumed to act as a full agonist of two types of melatonin receptors: melatonin receptor 1, with picomolar binding affinity, and melatonin receptor 2, with nanomolar binding affinity. Both receptors are part of the G-protein coupled receptors (GPCRs) family, specifically the Gi/o alpha subunit GPCRs.[13]
In vitro, melatonin functions as a high-capacity antioxidant or free radical scavenger, within mitochondria, playing a dual role in combating cellular oxidative stress.[13] First, it directly neutralizes free radicals, and second, it promotes the gene expression of essential antioxidant enzymes, such as superoxide dismutase, glutathione peroxidase, glutathione reductase, and catalase.[13] This increase in antioxidant enzyme expression is mediated through signal transduction pathways activated by the binding of melatonin to its receptors.[13] Through these mechanisms, melatonin is presumed to protect cells against oxidative stress in ways beyond regulating the sleep-wake cycle.[14][15][16]
Biological functions

Circadian rhythm
In mammals, melatonin is critical for the regulation of sleep–wake cycles, or circadian rhythms.[17] The establishment of regular melatonin levels in human infants occurs around the third month after birth, with peak concentrations observed between midnight and 8:00 am.[18] It has been documented that melatonin production diminishes as a person ages.[19] Additionally, a shift in the timing of melatonin secretion is observed during adolescence, resulting in delayed sleep and wake times, increasing their risk for delayed sleep phase disorder during this period.[20]
The antioxidant properties of melatonin were first recognized in 1993.[21] In vitro studies reveal that melatonin directly neutralizes various reactive oxygen species, including hydroxyl (OH•), superoxide (O2−•), and reactive nitrogen species such as nitric oxide (NO•).[22][23] In plants, melatonin works synergistically with other antioxidants, enhancing the overall effectiveness of each antioxidant.[23] This compound has been found to be twice as efficacious as vitamin E, a known potent lipophilic antioxidant, at scavenging peroxyl radicals.[24] The promotion of antioxidant enzyme expression, such as superoxide dismutase, glutathione peroxidase, glutathione reductase, and catalase, is mediated through melatonin receptor-triggered signal transduction pathways.[13]
Melatonin’s concentration in the mitochondrial matrix is significantly higher than that found in the blood plasma,[14][16] emphasizing its role not only in direct free radical scavenging but also in modulating the expression of antioxidant enzymes and maintaining mitochondrial integrity. This multifaceted role shows the physiological significance of melatonin as a mitochondrial antioxidant, a notion supported by numerous scholars.[14][16][15]
Furthermore, the interaction of melatonin with reactive oxygen and nitrogen species results in the formation of metabolites capable of reducing free radicals.[13][15] These metabolites, including cyclic 3-hydroxymelatonin, N1-acetyl-N2-formyl-5-methoxykynuramine (AFMK), and N1-acetyl-5-methoxykynuramine (AMK), contribute to the broader antioxidative effects of melatonin through further redox reactions with free radicals.[13][15]
Immune system
Melatonin’s interaction with the immune system is recognized, yet the specifics of these interactions remain inadequately defined.[25][26][27][28] An anti-inflammatory effect appears to be the most significant.[27][28] The efficacy of melatonin in disease treatment has been the subject of limited trials, with most available data deriving from small-scale, preliminary studies. It is posited that any beneficial immunological impact is attributable to melatonin’s action on high-affinity receptors (MT1 and MT2), which are present on immunocompetent cells. Preclinical investigations suggest that melatonin may augment cytokine production and promote the expansion of T cells,[29][30] thereby potentially mitigating acquired immunodeficiencies.[31]
Weight regulation
Melatonin’s potential to regulate weight gain is posited to involve its inhibitory effect on leptin, a hormone that serves as a long-term indicator of the body’s energy status.[32][33]
Biochemistry
Biosynthesis

The biosynthesis of melatonin in animals involves a sequence of enzymatic reactions starting with L-tryptophan, which can be synthesized through the shikimate pathway from chorismate, found in plants, or obtained from protein catabolism. The initial step in the melatonin biosynthesis pathway is the hydroxylation of L-tryptophan’s indole ring by the enzyme tryptophan hydroxylase, resulting in the formation of 5-hydroxytryptophan (5-HTP). Subsequently, 5-HTP undergoes decarboxylation, facilitated by pyridoxal phosphate and the enzyme 5-hydroxytryptophan decarboxylase, yielding serotonin.[34]
Serotonin, an essential neurotransmitter, is further converted into N-acetylserotonin by the action of serotonin N-acetyltransferase, using acetyl-CoA.[35] The final step in the pathway involves the methylation of N-acetylserotonin‘s hydroxyl group by hydroxyindole O-methyltransferase, with S-adenosyl methionine as the methyl donor, to produce melatonin.[35]
In bacteria, protists, fungi, and plants, the synthesis of melatonin also involves tryptophan as an intermediate but originates indirectly from the shikimate pathway. The pathway commences with D-erythrose 4-phosphate and phosphoenolpyruvate, and in photosynthetic cells, additionally involves carbon dioxide. While the subsequent biosynthetic reactions share similarities with those in animals, there are slight variations in the enzymes involved in the final stages.[36][37]
The hypothesis that melatonin synthesis occurs within mitochondria and chloroplasts suggests an evolutionary and functional significance of melatonin in cellular energy metabolism and defense mechanisms against oxidative stress, reflecting the molecule’s ancient origins and its multifaceted roles across different domains of life.[38]
Mechanism

The mechanism of melatonin biosynthesis initiates with the hydroxylation of L-tryptophan, a process that requires the cofactor tetrahydrobiopterin (THB) to react with oxygen and the active site iron of tryptophan hydroxylase. Although the complete mechanism is not entirely understood, two main mechanisms have been proposed:
The first mechanism involves a slow transfer of one electron from THB to molecular oxygen (O2), potentially producing a superoxide (O−2). This superoxide could then recombine with the THB radical to form 4a-peroxypterin. 4a-peroxypterin may either react with the active site iron (II) to create an iron-peroxypterin intermediate or directly transfer an oxygen atom to the iron, facilitating the hydroxylation of L-tryptophan.
Alternatively, the second mechanism proposes that oxygen interacts with the active site iron (II) first, forming iron (III) superoxide. This molecule could then react with THB to form an iron-peroxypterin intermediate.
Following the formation of iron (IV) oxide from the iron-peroxypterin intermediate, this oxide selectively attacks a double bond to yield a carbocation at the C5 position of the indole ring. A subsequent 1,2-shift of the hydrogen and the loss of one of the two hydrogen atoms on C5 would restore aromaticity, producing 5-hydroxy-L-tryptophan.[39]
The decarboxylation of 5-hydroxy-L-tryptophan to produce 5-hydroxytryptamine is then facilitated by a decarboxylase enzyme with pyridoxal phosphate (PLP) as a cofactor.[40] PLP forms an imine with the amino acid derivative, facilitating the breaking of the carbon–carbon bond and release of carbon dioxide. The protonation of the amine derived from tryptophan restores the aromaticity of the pyridine ring, leading to the production of 5-hydroxytryptamine and PLP.[41]
Serotonin N-acetyltransferase, with histidine residue His122, is hypothesized to deprotonate the primary amine of 5-hydroxytryptamine. This deprotonation allows the lone pair on the amine to attack acetyl-CoA, forming a tetrahedral intermediate. The thiol from coenzyme A then acts as a leaving group when attacked by a general base, producing N-acetylserotonin.[42]
The final step in the biosynthesis of melatonin involves the methylation of N-acetylserotonin at the hydroxyl position by SAM, resulting in the production of S-adenosyl homocysteine (SAH) and melatonin.[41][43]
Regulation
In vertebrates, the secretion of melatonin is regulated through the activation of the beta-1 adrenergic receptor by the hormone norepinephrine.[44] Norepinephrine increases the concentration of intracellular cAMP via beta-adrenergic receptors, which in turn activates the cAMP-dependent protein kinase A (PKA). PKA then phosphorylates arylalkylamine N-acetyltransferase (AANAT), the penultimate enzyme in the melatonin synthesis pathway. When exposed to daylight, noradrenergic stimulation ceases, leading to the immediate degradation of the protein by proteasomal proteolysis.[45]
Blue light, especially within the 460–480 nm range, inhibits the biosynthesis of melatonin,[46] with the degree of suppression being directly proportional to the intensity and duration of light exposure. Historically, humans in temperate climates experienced limited exposure to blue daylight during winter months, primarily receiving light from sources that emitted predominantly yellow light, such as fires.[47] The incandescent light bulbs used extensively throughout the 20th century emitted relatively low levels of blue light.[48] It has been found that light containing only wavelengths greater than 530 nm does not suppress melatonin under bright-light conditions.[49] The use of glasses that block blue light in the hours preceding bedtime can mitigate melatonin suppression.[50] Additionally, wearing blue-blocking goggles during the last hours before bedtime is recommended for individuals needing to adjust to an earlier bedtime since melatonin facilitates the onset of sleep.[51]
Metabolism
Melatonin is metabolized with an elimination half-life ranging from 20 to 50 minutes.[52][2][53] The primary metabolic pathway transforms melatonin into 6-hydroxymelatonin, which is then conjugated with sulfate and excreted in urine as a waste product.[54] It is primarily metabolized by the liver enzyme CYP1A2 and to a lesser extent by CYP1A1, CYP2C19, and CYP1B1.[54]
Measurement
For both research and clinical purposes, melatonin levels in humans can be determined through saliva or blood plasma analysis.[55]
Use as a medication and supplement
Insomnia
An extended-release pharmaceutical formulation of melatonin is approved under the brand name Circadin for the treatment of insomnia in certain settings, such as in people aged 55 years of age or older.[56][57][58][59] It is approved in the European Union, Israel, Australia, and countries in Asia and elsewhere in the world, but not in the United States (where it reached phase III clinical trials but was not approved).[58][59] The medication has been licensed since 2007.[58][59]
The 2023, European Insomnia Guideline recommended use of prolonged-release melatonin for treatment of insomnia in people age 55 year of age or older for up to three months.[60] It recommended against fast-release or over-the-counter melatonin for treatment of insomnia.[60] These recommendations were based on several meta-analyses published in 2022 and 2023.[60]
The American Academy of Sleep Medicine‘s 2017 clinical practice guidelines recommended against the use of melatonin in the treatment of insomnia due to poor effectiveness and very low quality of evidence.[61][62]
Circadian rhythm sleep disorders
Melatonin may be useful in the treatment of delayed sleep phase syndrome.[2]
Melatonin is known to reduce jet lag, especially in eastward travel. However, if it is not taken at the correct time, it can instead delay adaptation.[63]

Reviews
There are no reviews yet.