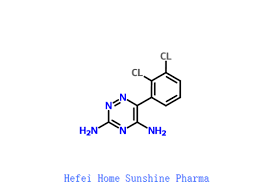
Buy Lamotrigine (Lamictal) Cas 84057-84-1
Lamotrigine (/ləˈmoʊtrɪˌdʒiːn/ luh-MOH-trih-jeen), sold under the brand name Lamictal among others, is a medication used to treat epilepsy and stabilize mood in bipolar disorder.[4][7] For epilepsy, this includes focal seizures, tonic-clonic seizures, and seizures in Lennox-Gastaut syndrome.[7] In bipolar disorder, lamotrigine has not been shown to reliably treat acute depression in any groups except for the severely depressed; but for patients with bipolar disorder who are not currently symptomatic, it appears to reduce the risk of future episodes of depression.[8] Lamotrigine is also used off label for unipolar depression (major depressive disorder) and depersonalization-derealization disorder.[9]
Common side effects include nausea, sleepiness, headache, vomiting, trouble with coordination, and rash.[7] Serious side effects include excessive breakdown of red blood cells, increased risk of suicide, severe skin reaction (Stevens–Johnson syndrome), and allergic reactions, which can be fatal.[7] Lamotrigine is a phenyltriazine,[4] making it chemically different from other anticonvulsants.[7] Its mechanism of action is not clear, but it appears to inhibit release of excitatory neurotransmitters via voltage-sensitive sodium channels and voltage-gated calcium channels in neurons.[7][10][11]
Lamotrigine was first marketed in Ireland in 1991,[12] and approved for use in the United States in 1994.[7][13] It is on the World Health Organization’s List of Essential Medicines.[14] In 2023, it was the most commonly prescribed mood stabilizer and 59th most commonly prescribed medication in the United States, with more than 10 million prescriptions.[15][16]
Medical uses
Epilepsy
Lamotrigine is considered a first-line drug for primary generalized tonic-clonic seizures (includes simple partial, complex partial, and secondarily generalized seizures such as focal-onset tonic-clonic seizures). It is also used as an alternative or adjuvant medication for partial seizures, such as absence seizure, myoclonic seizure, and atonic seizures.[17][18] The evidence supporting the use of lamotrigine as an add-on therapy for drug-resistant generalized tonic-clonic seizures is not clear enough to inform clinical practice.[19] Although low-certainty evidence suggests that it reduces generalized tonic-clonic seizures by 50%, the level of uncertainty indicates that the actual findings could be significantly different.[19] Evidence supporting the use of lamotrigine as an add-on therapy for drug-resistant focal epilepsy found that it is likely effective for reducing seizure frequency and is generally well tolerated.[20] Lamotrigine has some side effects including a risk of dizziness, nausea, ataxia, or visual disturbances such as diplopia.[20] The long-term effects of lamotrigine have not been investigated.[20]
Lennox–Gastaut syndrome
Lamotrigine is one of a small number of FDA-approved therapies for the form of epilepsy known as Lennox–Gastaut syndrome.[21] It reduces the frequency of LGS seizures, and is one of two medications known to decrease the severity of drop attacks.[22] Combination with valproate is common, but this increases the risk of Stevens–Johnson syndrome, and necessitates reduced dosing due to the interaction of these drugs.[23]

Reviews
There are no reviews yet.