Buy Ethchlorvynol (Pfizer) Cas 113-18-8
Ethchlorvynol is a GABA-ergic hypnotic sedative medication first developed by Pfizer in the 1950s.[4] In the United States, it was sold by Abbott Laboratories under the trade name Placidyl.[2] Placidyl was available in 200 mg, 500 mg, and 750 mg strength gel filled capsules. While the 500 mg and 750 mg strength capsules were for use in reducing sleep latency, the 200 mg strength capsules were intended to be used to re-induce sleep in case of early awakening. Abbott discontinued production in 1999 due to problems of the widespread abuse of minor tranquilizers, and non-barbiturate sedatives largely being replaced by the benzodiazepine family (with drugs such as Librium and Valium);[5][2] by the end of 2001, ethchlorvynol reserves had dried up in the United States.
Theoretically, ethchlorvynol could be manufactured for sale in the United States by another pharmaceutical company (subject to FDA approval of such manufacture), though no pharmaceutical company has chosen to do so. Individuals with a valid prescription for the substance may legally transport a reasonable amount of ethchlorvynol with them into the United States. The availability of ethchlorvynol resembles similar barbiturate-like, GABA-ergic sedatives, such as glutethimide (Doriden, which ceased production in U.S. by 1993), ethinamate (Valmid), and methyprylon (vended as Noludar, Noctan, and Dimerin), which were voluntarily withdrawn in favor of benzodiazepine hypnotics in 1965, and supplies widely eliminated by 1969. These are all controlled substances, but are not illegal.
Use and effects
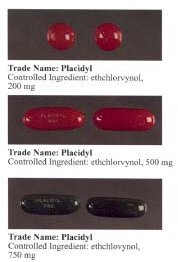
Ethchlorvynol was indicated to treat insomnia, and was widely prescribed in the 1960s and 1970s; prescriptions had fallen significantly by 1990, however, as other hypnotics that were considered safer and less dangerous in overdose became more common.
Availability
Ethchlorvynol is no longer prescribed in the United States due to unavailability, but is still available in some countries, and would still be considered legal to possess and use with a valid prescription.
Adverse effects
Along with expected sedative effects (e.g. relaxation and drowsiness), reported adverse effects of ethchlorvynol include gastrointestinal upset (including nausea and vomiting), dizziness, blurred or altered vision, numbness or tingling, and unsteadiness or impaired coordination. Hypersensitivity reactions such as skin rash have also been reported.[6]
Misuse by injection has been associated with serious acute toxicity. Reports describe severe noncardiogenic pulmonary oedema and haemodynamic effects following intravenous injection of ethchlorvynol (Placidyl), and public health sources have warned of cardiovascular or pulmonary injury arising from improper intravenous or intra-arterial injection of the drug.[7][8]

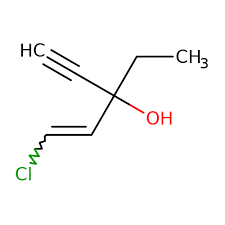
Reviews
There are no reviews yet.