Buy Dextropropoxyphene Cas 469-62-5
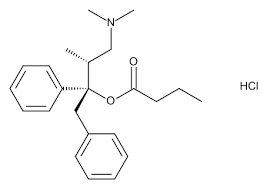
Dextropropoxyphene[5] is an opioid analgesic patented in 1955[6] and manufactured by Eli Lilly and Company. It is an optical isomer of levopropoxyphene. It is intended to treat mild pain and also has antitussive (cough suppressant) and local anesthetic effects. The drug has been taken off the market in Europe and the United States due to concerns of fatal overdoses and heart arrhythmias.[7] It is still available in Australia, albeit with restrictions after an application by its manufacturer to review its proposed banning.[8] Its onset of analgesia (pain relief) is said to be 20–30 minutes and peak effects are seen about 1.5–2.0 hours after oral administration.[3]
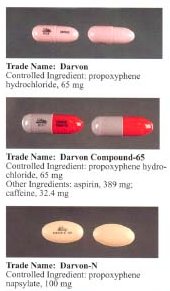
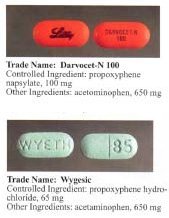
Dextropropoxyphene is sometimes combined with paracetamol (acetaminophen). Trade names include Darvocet-N, Di-Gesic,[9] and Darvon with APAP (for dextropropoxyphene and paracetamol).[10] The British approved name (i.e. the generic name of the active ingredient) of the paracetamol/dextropropoxyphene preparation is co-proxamol (sold under a variety of brand names); however, it has been withdrawn since 2007, and is no longer available to new patients, with exceptions.[11] The paracetamol combinations are known as Capadex or Di-Gesic in Australia, Lentogesic in South Africa, and Di-Antalvic in France (unlike co-proxamol, which is an approved name, these are all brand names).
Medical uses
Analgesia
Dextropropoxyphene is generally considered a weak analgesic, with several studies finding its efficacy is no better than acetaminophen.[12] Like codeine, it is a weak opioid. However, dextropropoxyphene has one-third to one-half of the analgesic activity of codeine.[12]
Restless legs syndrome
Dextropropoxyphene has been found to be helpful in relieving the symptoms of restless legs syndrome.[13][14][15]
Available forms
Propoxyphene was initially introduced as propoxyphene hydrochloride. Shortly before the patent on propoxyphene expired, propoxyphene napsylate form was introduced to the market. The napsylate salt (the salt of naphthalene-2-sulfonic acid) is claimed to be less prone to non-medical use, because it is almost insoluble in water, so cannot be used for injection. Napsylate also gives lower peak blood level.[16] Because of different molar mass, a dose of 100 mg of propoxyphene napsylate is required to supply an amount of propoxyphene equivalent to that present in 65 mg of propoxyphene hydrochloride.
Before the FDA-directed recall, dextropropoxyphene HCl was available in the United States as a prescription formulation combined with paracetamol (acetaminophen) in ratio from 30 mg / 600 mg to 100 mg / 650 mg (or 100 mg / 325 mg in the case of Balacet), respectively, usually named Darvocet.
In Australia, dextropropoxyphene is available on prescription, both as a combined product (32.5 mg dextropropoxyphene per 325 mg paracetamol branded as Di-gesic, Capadex, or Paradex; it is also available in pure form (100 mg capsules) known as Doloxene, however its use has been restricted.[8]
Contraindications
Dextropropoxyphene is contraindicated in patients allergic to paracetamol (acetaminophen) or dextropropoxyphene, and in alcoholics. It is not intended for use in patients who are prone to suicide, anxiety, panic, or addiction.
Side effects
Severe toxicity can occur with small increments above the therapeutic dose including cardiotoxicity, and fatal overdoses. This is especially true when the drug is combined with alcohol.[17] Other side effects include:[18]
- Constipation
- Itching
- Drowsiness
- Nausea
- Sensorineural deafness
Pharmacology
Dextropropoxyphene acts as a μ-opioid receptor agonist. It also acts as a potent, noncompetitive α3β4 neuronal nicotinic acetylcholine receptor antagonist,[19] as well as a weak serotonin reuptake inhibitor.
Toxicity
Overdose is commonly broken into two categories – liver toxicity (from paracetamol poisoning) and dextropropoxyphene overdose. It may also account for mood– or thought-altering effects.
An overdose of dextropropoxyphene may lead to various systemic effects. Excessive opioid receptor stimulation is responsible for the CNS depression, respiratory depression, aspiration pneumonia, miosis, and gastrointestinal effects seen in propoxyphene poisoning. In the presence of amphetamine, propoxyphene overdose increases CNS stimulation and may cause fatal convulsive seizures.[20]
In addition, both propoxyphene and its metabolite norpropoxyphene have local anesthetic effects at concentrations about 10 times those necessary for opioid effects. Norpropoxyphene is a more potent local anesthetic than propoxyphene, and they are both more potent than lidocaine.[21] Local anesthetic activity appears to be responsible for the arrhythmias and cardiovascular depression seen in propoxyphene poisoning.[22]
Both propoxyphene and norpropoxyphene are potent blockers of cardiac membrane sodium channels, and are more potent than lidocaine, quinidine, and procainamide in this respect.[23] As a result, propoxyphene and norpropoxyphene appear to have the characteristics of a Vaughn-Williams Class Ic antiarrhythmic.
These direct cardiac effects include decreased heart rate (i.e. cardiovascular depression), decreased contractility, and decreased electrical conductivity (i.e., increased PR, AH, HV, and QRS intervals). These effects appear to be due to their local anesthetic activity and are not reversed by naloxone.[21][22][24] Widening of the QRS complex appears to be a result of a quinidine-like effect of propoxyphene, and sodium bicarbonate therapy appears to have a positive direct effect on the QRS dysrhythmia.[25]
Seizures may result from either opioid or local anesthetic effects.[21] Pulmonary edema may result from direct pulmonary toxicity, neurogenic/anoxic effects, or cardiovascular depression.[22]
Balance disorder is possible, with risk of falls from standing height.
History
|
This section needs expansion. You can help by adding missing information. (September 2025)
|
Dextropropoxyphene was successfully tested in 1954 as part of US Navy and CIA-funded research on nonaddictive substitutes for codeine.[26]
Use in organic synthesis
Without the propionyl group on the oxygen, the non-esterified alcohol precursor of propoxyphene (both enantiomers, known as darvon alcohol and novrad alcohol) have been employed as stoichiometric chiral reagents for asymmetric carbonyl reduction reactions involving aluminium hydride reagents.[27][28]

Reviews
There are no reviews yet.