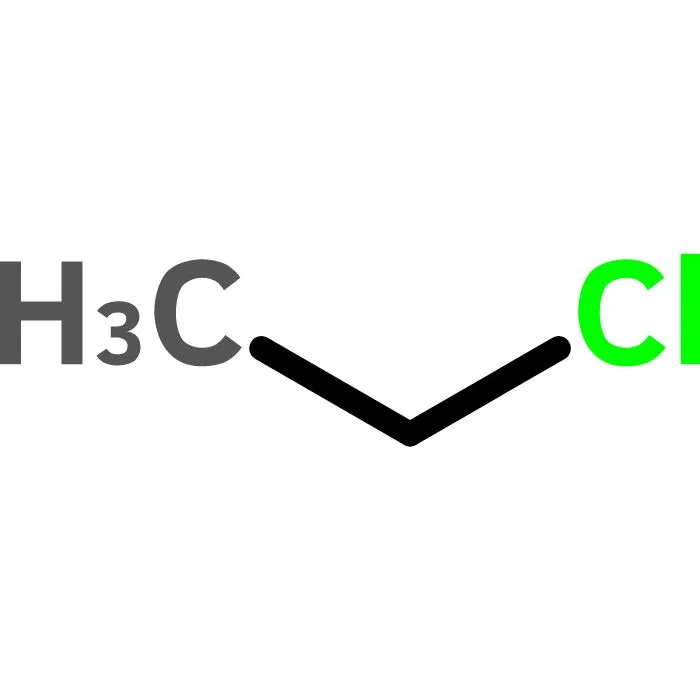
Buy Chloroethane (ethyl chloride) Cas 75-00-3
Chloroethane, commonly known as ethyl chloride, is a chemical compound with chemical formula CH3CH2Cl, once widely used in producing tetraethyllead, a gasoline additive. It is a colorless, flammable gas or refrigerated liquid with a faintly sweet odor.[11]
Ethyl chloride was first synthesized by Basil Valentine by reacting ethanol and hydrochloric acid in 1440.[11] Glauber made it in 1648 by reacting ethanol and zinc chloride.[11]
Production
Chloroethane is produced by hydrochlorination of ethylene:[11]
- C2H4 + HCl → C2H5Cl
At various times in the past, chloroethane has also been produced from ethanol and hydrochloric acid, from ethane and chlorine, or from ethanol and phosphorus trichloride, but these routes are no longer economical. Some chloroethane is generated as a byproduct of polyvinyl chloride production.
Uses
Chloroethane is an inexpensive ethylating agent. It reacts with aluminium metal to give ethylaluminium sesquichloride, a precursor to polymers and other useful organoaluminium compounds.[12] Chloroethane is used to convert cellulose to ethylcellulose, a thickening agent and binder in paints, cosmetics, and similar products.
Like other chlorinated hydrocarbons, chloroethane has been used as a refrigerant, an aerosol spray propellant, an anesthetic, and a blowing agent for foam packaging. For a time it was used as a promoter chemical in the aluminium chloride catalyzed process to produce ethylbenzene, the precursor for styrene monomer. At present though, it is not widely used in any of these roles.
Obsolete uses
Beginning in 1922 and continuing through most of the 20th century, the major use of chloroethane was to produce tetraethyllead (TEL), an anti-knock additive for gasoline. TEL has been or is being phased out in most of the industrialized world, and the demand for chloroethane has fallen sharply.[11]
Niche uses

Chloroethane has a low boiling point, so when applied topically, the heat absorbed by the boiling liquid produces a deep and rapid chill. When sprayed on the skin, this chill has a mild anesthetic effect, which can be useful when removing splinters or incising abscesses in a clinical setting. Chloroethane was standard equipment in casualty wards.[when?] It was commonly used to induce general anaesthesia before continuing with diethyl ether, which has a much slower uptake.[citation needed] In dentistry, chloroethane is used as one of the means of diagnosing a ‘dead tooth’, i.e., one in which the pulp has died. A small amount of the substance is placed on the suspect tooth using a cotton wad; if the tooth is still alive this should be sensed by the patient as mild discomfort that subsides when the wad is removed.[citation needed]

Reviews
There are no reviews yet.