Buy Butorphanol (morphinan) Cas 42408-82-2
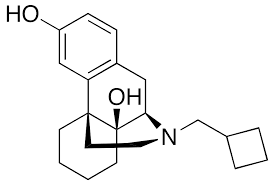
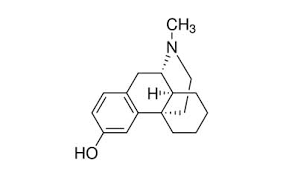
Butorphanol is a morphinan-type synthetic agonist–antagonist opioid analgesic developed by Bristol-Myers.[2][3][4] Butorphanol is most closely structurally related to levorphanol.[citation needed] Butorphanol is available as the tartrate salt in injectable, tablet, and intranasal spray formulations. The tablet form is only used in dogs, cats and horses due to low bioavailability in humans.
It was patented in 1971 and approved for medical use in 1979.[5]
Medical uses
The most common indication for butorphanol is management of migraine using the intranasal spray formulation. It may also be used parenterally for management of moderate-to-severe pain, as a supplement for balanced general anesthesia, and management of pain during labor. Butorphanol is also quite effective at reducing post-operative shivering (owing to its kappa agonist activity). Butorphanol is more effective in reducing pain in women than in men.[6]
Pharmacology
Butorphanol exhibits partial agonist and antagonist activity at the μ-opioid receptor, as well as partial agonist activity at the κ-opioid receptor (Ki = 2.5 nM; EC50 = 57 nM; Emax = 57%).[6][7] Stimulation of these receptors on central nervous system neurons causes an intracellular inhibition of adenylate cyclase, closing of influx membrane calcium channels, and opening of membrane potassium channels. This leads to hyperpolarization of the cell membrane potential and suppression of action potential transmission of ascending pain pathways. Because of its κ-agonist activity, at analgesic doses butorphanol increases pulmonary arterial pressure and cardiac work. Additionally, κ-agonism can cause dysphoria at therapeutic or supratherapeutic doses; this gives butorphanol a lower potential for abuse than other opioid drugs.[8]
Society and culture
Name
Within the INN, USAN, BAN, and AAN naming systems this drug is known as butorphanol, while within JAN it is named torbugesic.[2][3][4] As the tartrate salt, butorphanol is known as butorphanol tartrate (USAN, BAN).[2][3][4]
Legal status
Butorphanol is listed under the Single Convention on Narcotic Drugs 1961 and in the United States is a Schedule IV controlled substance with a DEA ACSCN of 9720.[9] The free base conversion ratio of the hydrochloride is 0.69.[10] Butorphanol was originally a Schedule II controlled substance and was later decontrolled at one point.[citation needed]
Veterinary use
In veterinary anesthesia, butorphanol (trade name: Torbugesic) is widely used as a sedative and analgesic in dogs, cats and horses. For sedation, it may be combined with tranquilizers such as alpha-2 agonists (medetomidine), benzodiazepines, or acepromazine in dogs, cats and exotic animals. It is frequently combined with xylazine or detomidine in horses.[11] Butorphanol may be administered intravenously, intramuscularly, subcutaneously, or per os. Intramuscular and subcutaneous administration may cause pain. Oral tablets have poor bioavailability and are not suitable for analgesia. Instranasal usage has been reported in parrots and rabbits.[12] Butorphanol when administered at 0.4 mg/kg given IV/IM does not provide sufficient post-operative analgesia for laparotomy and shoulder arthrotomy in dogs and ovariohysterectomy in bitches.[12][13][14][15] When butorphanol is used as a sedative in dogs—either on its own or with dexmedetomidine—it provides fast sedation and is faster than sedation with methadone.[12][16][17] Butorphanol is also approved as an antitussive in the dog.[12]

Reviews
There are no reviews yet.