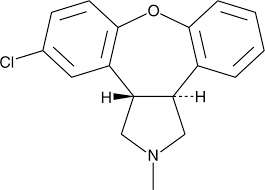
Buy Asenapine (Saphris) Cas 65576-45-6
Asenapine, sold under the brand name Saphris among others, is an atypical antipsychotic medication used to treat schizophrenia and acute mania associated with bipolar disorder as well as the medium to long-term management of bipolar disorder.[3][9]
It was chemically derived via altering the chemical structure of the tetracyclic (atypical) antidepressant, mianserin.[10]
It was initially approved in the United States in 2009[11] and approved as a generic medication in 2020.[12]
Medical uses
Asenapine has been approved by the FDA for the acute treatment of adults with schizophrenia and acute treatment of manic or mixed episodes associated with bipolar I disorder with or without psychotic features in adults.[11] In Australia asenapine’s approved (and also listed on the PBS) indications include the following:[13]
- Schizophrenia
- Treatment, for up to 6 months, of an episode of acute mania or mixed episodes associated with bipolar I disorder
- Maintenance treatment, as monotherapy, of bipolar I disorder
In the European Union and the United Kingdom, asenapine is only licensed for use as a treatment for acute mania in bipolar I disorder.[2][6][3]
Asenapine is absorbed readily if administered sublingually, but is poorly absorbed when swallowed.[14] A transdermal formulation of asenapine was approved in the United States in October 2019 under the brand name Secuado.[7]
Schizophrenia
A Cochrane systematic review found that while Asenapine has some preliminary evidence that it improves positive, negative, and depressive symptoms, it does not have enough research to merit a certain recommendation of asenapine for the treatment of schizophrenia.[15]
Bipolar disorder
For the medium-term and long-term management and control of both depressive and manic features of bipolar disorder asenapine was found to be equally effective as olanzapine, but with a substantially superior side effect profile.[9]
In acute mania, asenapine was found to be significantly superior to placebo.[9] As for its efficacy in the treatment of acute mania, a recent meta-analysis showed that it produces comparatively small improvements in manic symptoms in patients with acute mania and mixed episodes than most other antipsychotic drugs such as risperidone and olanzapine (with the exception of ziprasidone). Drop-out rates (in clinical trials) were also unusually high with asenapine.[16] According to a post-hoc analysis of two 3-week clinical trials it may possess some antidepressant effects in patients with acute mania or mixed episodes.[17]
Adverse effects
Adverse effect incidence[4][5][2][6]
Very common (>10% incidence) adverse effects include:
Common (1–10% incidence) adverse effects include:
- Weight gain†
- Increased appetite
- Extrapyramidal side effects (EPS; such as dystonia, akathisia, dyskinesia, muscle rigidity, parkinsonism)
- Sedation
- Dizziness
- Dysgeusia (altered taste)
- Oral hypoaesthesia (numbness), only when taken sublingually. Transdermal asenapine was shown to eliminate this side effect.[7]
- Increased alanine aminotransferase
- Dyspepsia, stomach discomfort, and/or vomiting[a]
- Fatigue

Reviews
There are no reviews yet.