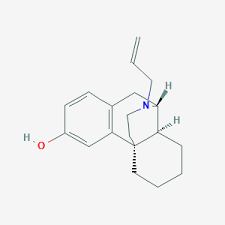
Buy Dextrallorphan (DXA) Cas 5822-43-5
Dextrallorphan (DXA) is a chemical of the morphinan class that is used in scientific research. It acts as a σ1 receptor agonist and NMDA receptor antagonist.[1][2][3][4] It has no significant affinity for the σ2, μ-opioid, or δ-opioid receptor, or for the serotonin or norepinephrine transporter.[2][5] As an NMDA receptor antagonist, in vivo, it is approximately twice as potent as dextromethorphan, and five-fold less potent than dextrorphan.[3]
Uses in Scientific Research
Masking of sigma-1 receptor
Dextrallorphan is often used in research to block σ1 receptor sites so that σ2 receptor sites (which have not been cloned yet [when?]) can be studied.[6][7][8] It was hypothesized that both of these sigma (σ) receptors were opioid receptors, due to their affinity for psychoactive drugs. However, it is now understood that they are non-opioid receptors that bind to certain psychoactive drugs, like dextrallorphan.[9] One example of dextrallorphan being used to mask σ1 receptor sites was seen in a study on the localization of the σ2 receptor in detergent-resistant lipid raft domains.[6] It has also been used to mask σ1 receptor sites so that σ2 receptor binding characteristics in the rat liver could be determined, by labeling σ2 receptor sites with [3H]l,3-di-o-tolylguanidine (DTG) in the presence of 1 μM dextrallorphan solution.[8]
Animal Studies
Dextrallorphan was used in Spraque-Dawley rats to study cerebellar Purkinje neurons electrophysical responses to the drug when it was applied iontophoretically as a sigma (σ) receptor ligand. Dextrallorphan increased the firing rate by 14%, suggesting that sigma (σ) ligands (like dextrallorphan) alter the spontaneous firing of Purkinje neurons and cause motor effects.[10]
In another study, dextrallorphan, along with other opioid derivatives, was found to be a potent inhibitor of etorphine-inaccessible (EI) sites in the guinea-pig brain. Dextrallorphan was of the top three most potent opioid inhibitors of those studied, with a concentration of 67 nM required to show 50% inhibition.[1]

Reviews
There are no reviews yet.