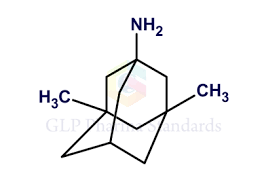
Buy Memantine (Namenda) Cas 19982-08-2
Memantine, sold under the brand name Namenda among others, is a medication used to slow the progression of moderate-to-severe Alzheimer’s disease.[10][11][8] It is taken by mouth.[10][8]
Common side effects include headache, constipation, sleepiness, and dizziness.[10][11] Severe side effects may include blood clots, psychosis, and heart failure.[11] It is believed to work by acting on NMDA receptors, working as a pore blocker of these ion channels.[8][10]
Memantine was first discovered in 1963.[8][12][13] It was approved for medical use in Germany in 1989, in the European Union in 2002, and in the United States in 2003.[13][10][14] It is available as a generic medication.[11] In 2023, it was the 145th most commonly prescribed medication in the United States, with more than 3 million prescriptions.[15][16]
Medical uses
Alzheimer’s disease and dementia
Memantine is used to treat moderate-to-severe Alzheimer’s disease, especially for people who are intolerant of or have a contraindication to AChE (acetylcholinesterase) inhibitors.[17][18] One guideline recommends memantine or an AChE inhibitor be considered in people in the early-to-mid stage of dementia.[19]
Memantine has been associated with a modest improvement;[20] with small positive effects on cognition, mood, behavior, and the ability to perform daily activities in moderate-to-severe Alzheimer’s disease.[21][22] There does not appear to be any benefit in mild disease.[23]
Memantine when added to donepezil in those with moderate-to-severe dementia resulted in “limited improvements” in a 2017 review.[24] The UK National Institute for Clinical Excellence (NICE) issued guidance in 2018 recommending consideration of the combination of memantine with donepezil in those with moderate-to-severe dementia.[25]
Radiation therapy
Memantine has been recommended for use by professional organization consensus to prevent neurocognitive decline after whole brain radiotherapy.[26]
Adverse effects
Memantine is, in general, well tolerated.[20] Common adverse drug reactions (≥1% of people) include confusion, dizziness, drowsiness, headache, insomnia, agitation, and/or hallucinations. Less common adverse effects include vomiting, anxiety, hypertonia, cystitis, and increased libido.[20][27]
Like many other NMDA receptor antagonists, memantine behaves as a dissociative anesthetic at supratherapeutic doses.[28] Despite isolated reports, recreational use of memantine is rare due to the drug’s long duration and limited availability.[28] Additionally, memantine seems to lack effects such as euphoria or hallucinations.[29]
Memantine appears to be generally well tolerated by children with autism spectrum disorder.[30]
Pharmacology
Pharmacodynamics
Memantine is an uncompetitive NMDA receptor antagonist, with an IC50 binding affinity of ~500-1,000 nM (0.5-1 μM),[31] but displays higher potency at extrasynaptic NMDA receptors (IC50 = ~22 nM),[31] suggesting preferential inhibition of pathological glutamatergic signaling.
Memantine also exhibits activity at several other

Reviews
There are no reviews yet.