Buy Nefazodone (Serzone) Cas 83366-66-9
Nefazodone, sold formerly under the brand names Serzone, Dutonin, and Nefadar among others, is an atypical antidepressant medication which is used in the treatment of depression and for other uses.[4][5][6][7] Nefazodone was withdrawn in most countries by 2004[8][9] (due to liver toxicity[10]). On December 2021, it was published that despite shortage it is still available in the United States.[11]As of February 2026 it is available in the United States in generic form. The medication is taken by mouth.[10]
Side effects of nefazodone include dry mouth, sleepiness, nausea, dizziness, blurred vision, weakness, lightheadedness, confusion, and postural low blood pressure, among others.[10] Rarely, nefazodone can cause serious liver damage, with an incidence of death or liver transplantation of about 1 in every 250,000 to 300,000 patient years.[10] Nefazodone is a phenylpiperazine compound and is related to trazodone. It has been described as a serotonin antagonist and reuptake inhibitor (SARI) due to its combined actions as a potent antagonist of the serotonin 5-HT2A and 5-HT2C receptors and weak serotonin–norepinephrine–dopamine reuptake inhibitor (SNDRI).
Nefazodone was introduced for medical use in 1994.[7][12][8] Generic versions were introduced in 2003.[13] Serious liver toxicity was first reported with nefazodone in 1998, and it was withdrawn from most markets by 2004.[8][9] However, as of 2023, it continues to be available in the United States in generic from one manufacturer, Teva Pharmaceuticals[citation needed][14][failed verification] and is manufactured in Israel.[15]
Medical uses
Nefazodone is used to treat major depressive disorder, aggressive behavior, anxiety,[16] and panic disorder.[17]
Available forms
Nefazodone is available as 50 mg, 100 mg, 150 mg, 200 mg, and 250 mg tablets for oral ingestion.[18]
Contraindications
Contraindications include the coadministration of terfenadine, astemizole, cisapride, pimozide, or carbamazepine. Nefazodone is contraindicated in patients who were withdrawn from nefazodone because of evident liver injury as well as those that have shown hypersensitivity to the drug, its inactive ingredients, or other phenylpiperazine antidepressants. Furthermore, the coadministration of triazolam and nefazodone should be avoided for all patients, including the elderly, since it causes a significant increase in the plasma level of triazolam and not all commercially available dosage forms of triazolam permit a sufficient dosage reduction. If coadministrated, a 75% reduction in the initial dosage of triazolam is recommended.[18]
Side effects
Common and mild side effects of nefazodone reported in clinical trials more often than placebo include dry mouth (25%), sleepiness (25%), nausea (22%), dizziness (17%), blurred vision (16%), weakness (11%), lightheadedness (10%), confusion (7%), and orthostatic hypotension (5%). Rare and serious adverse reactions may include allergic reactions, fainting, painful/prolonged erection, and jaundice.[10] Nefazodone is not especially associated with increased appetite and weight gain.[19] It is also known for having low levels of sexual side effects in comparisons to SSRIs.[20][21]

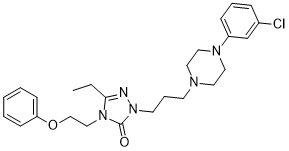
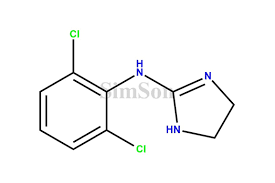
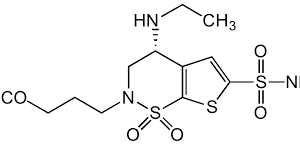
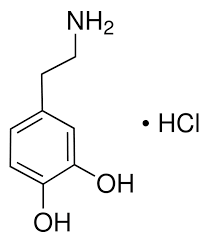
Reviews
There are no reviews yet.