Buy Chloroform (trichloromethane) Cas 67-66-3
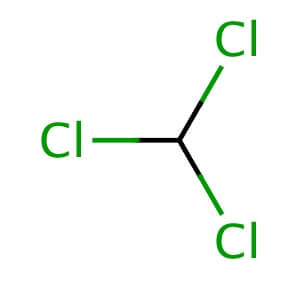
Chloroform,[9] or trichloromethane (often abbreviated as TCM), is an organochloride with the formula CHCl3 and a common solvent. It is a volatile, colorless, sweet-smelling, dense liquid produced on a large scale as a precursor to refrigerants and polytetrafluoroethylene (PTFE).[10] Chloroform was once used as an inhalational anesthetic between the 19th century and the first half of the 20th century.[11][12] It is miscible with many solvents but it is only very slightly soluble in water (only 8 g/L at 20°C).
Structure and name
The molecule adopts a tetrahedral molecular geometry with C3v symmetry.[13] The chloroform molecule can be viewed as a methane molecule with three hydrogen atoms replaced with three chlorine atoms, leaving a single hydrogen atom.
The name “chloroform” is a portmanteau of terchloride (tertiary chloride, a trichloride) and formyle, an obsolete name for the methylylidene radical (CH) derived from formic acid.[14]
Natural occurrence
Many kinds of seaweed produce chloroform, and fungi are believed to produce chloroform in soil.[15] Abiotic processes are also believed to contribute to natural chloroform productions in soils, although the mechanism is still unclear.[16]
History
Chloroform was synthesized independently by several investigators c. 1831:
- Moldenhawer, a German pharmacist from Frankfurt an der Oder, appears to have produced chloroform in 1830 by mixing chlorinated lime with ethanol; however, he mistook it for Chloräther (chloric ether, 1,2-dichloroethane).[17][18]
- Samuel Guthrie, a U.S. physician from Sackets Harbor, New York, also appears to have produced chloroform in 1831 by reacting chlorinated lime with ethanol, and noted its anaesthetic properties; however, he also believed that he had prepared chloric ether.[19][20][21]
- Justus von Liebig carried out the alkaline cleavage of chloral. Liebig incorrectly states that the empirical formula of chloroform was C2Cl5 and named it “Chlorkohlenstoff” (“carbon chloride”).[22][23]
- Eugène Soubeiran obtained the compound by the action of chlorine bleach on both ethanol and acetone.[24]
In 1834, French chemist Jean-Baptiste Dumas determined chloroform’s empirical formula and named it:[25] “Es scheint mir also erweisen, dass die von mir analysirte Substanz, … zur Formel hat: C2H2Cl6.” (Thus it seems to me to show that the substance I analyzed … has as [its empirical] formula: C2H2Cl6.). [Note: The coefficients of his empirical formula should be halved.] … “Diess hat mich veranlasst diese Substanz mit dem Namen ‘Chloroform’ zu belegen.” (This had caused me to impose the name “chloroform” upon this substance [i.e., formyl chloride or chloride of formic acid].)
In 1835, Dumas prepared the substance by alkaline cleavage of trichloroacetic acid.
In 1842, Robert Mortimer Glover in London discovered the anaesthetic qualities of chloroform on laboratory animals.[26]
In 1847, Scottish obstetrician James Y. Simpson was the first to demonstrate the anaesthetic properties of chloroform (provided by local pharmacist William Flockhart of Duncan, Flockhart and company,[27]) in humans, and helped to popularize the drug for use in medicine.[28]
The application of chloroform remained dangerous, and many deaths occurred through accidental overdose.[29][30] In 1848, John Snow developed an inhaler that regulated the dosage.[29]
By the 1850s, chloroform was being produced on a commercial basis.[30] An apparatus that could apply it safely and controllably was invented by Joseph Thomas Clover in 1862.[31][32]
In Britain, about 750,000 doses a week were being produced by 1895,[30] using the Liebig procedure, which retained its importance until the 1960s. Today, chloroform – along with dichloromethane – is prepared exclusively and on a massive scale by the chlorination of methane and chloromethane.[10]


Reviews
There are no reviews yet.